New mechanism of antibiotic tetracycline discovered
8 March 2021
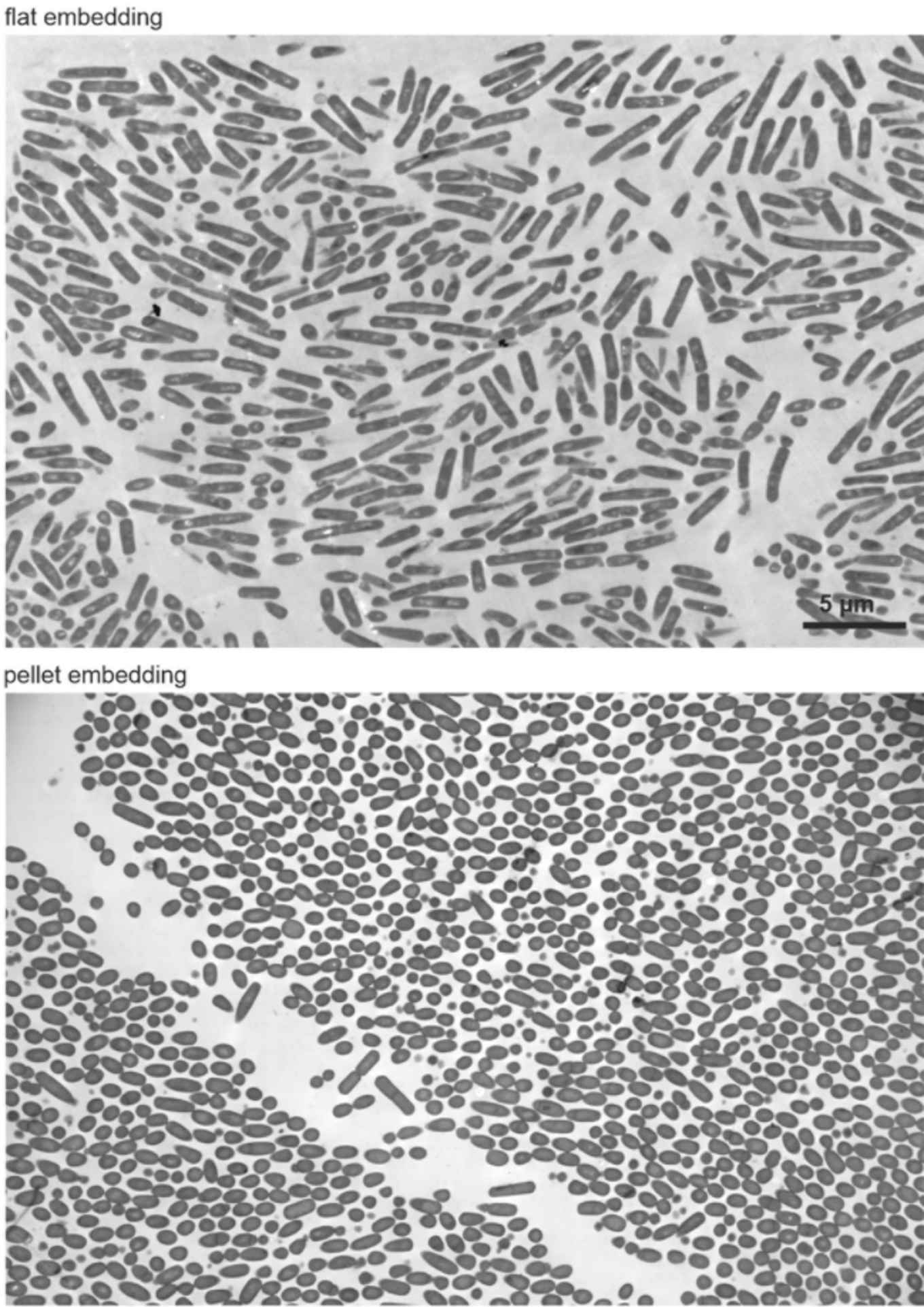
The team consitst of microbiologists from the University of Amsterdam’s Swammerdam Institute for Life Sciences, in collaboration with scientists from the AUMC, VU and the Chalmers University of Technology in Gothenburg. The group was led by Michaela Wenzel and Leendert Hamoen (UvA). They were working on an improved technique to check bacteria by electron microscopy in a quantifiable manner. To test their technique, they measured the morphological reaction of bacteria treated with different antibiotics. Surprisingly, while doing so, they found a new mechanism of how the well-known antibiotic tetracycline fights bacterial growth.
Broadly used antibiotic
Tetracyclines are one of the most commonly used antibiotic groups in both human and veterinary medicine. Tetracycline serves as effective antibiotic against a wide range of bacteria. Its most known mechanism of action is prohibiting bacterial growth and survival by blocking bacterial ribosomes, the structures that orchestrate protein translation. These ribosomes are present in all bacteria, and therefore tetracycline proved widely successful. However, this success comes with a downside. Bacteria can become resistant against antibiotics, posing a threat to treatment options. As for most antibiotics, this was also found for tetracycline.
By understanding exactly which mechanisms antibiotics use to block bacterial growth, we can learn how bacteria adapt to cope with these mechanisms upon becoming resistant. That is why fundamental research on how bacteria grow, and what happens when they are exposed to antibiotics, remains important. There is still a lot to be learned about this. This knowledge contributes to the prevention of resistance, and hopefully it also helps us to find clues for the discovery and development of new molecules with antibiotic activity.
Novel protocol
The goal of the study was to optimize a preparation technique to more efficiently study bacteria via transmission electron microscopy (TEM). TEM is a powerful microscopy technique that allows researchers to examine the morphology and ultrastructure of bacteria and other very small cells. While the technique has been used to visualize bacteria for over 60 years, the way of preparing a sample has remained unchanged. Wenzel and colleagues wanted to improve this, because the current technique has an important limitation: that there is no control on how the bacteria are oriented.
When studying rod-shaped bacteria, this meant that they had to spend a lot of time and sample to search for bacteria that lay oriented in such a way that they can see the complete long side of the bacteria. They optimized a protocol to prepare a sample in such a way that all bacteria get embedded on a flat surface, making all bacteria lay in the correct orientation under the microscope. This drastically improves how many bacteria can be imaged per experiment, while needing less sample and less time to find them.
Mechanism of tetracycline
To assess the potential of the new technique, the researchers monitored the reaction of Bacillus subtilis, a well-known model strain often used in the lab, to tetracycline. Surprisingly, they found that it causes deformations in the membrane of the bacteria. They dived into the details behind this and found that it was not the result of blocking the ribosomes, but a different mechanism was causing the deformations. The researchers examined the localization of three different proteins that localize in the membrane of healthy bacteria by fluorescent labelling. They found that tetracycline leads to these proteins getting wrongly localized on the membrane. This happens independently of the blocking of ribosomes, and therefore turns out to be a newly discovered mechanism of tetracycline.
Resistance against tetracycline occurs relatively slowly, while this is not the case for other antibiotics targeting ribosomes. It was not entirely clear why this was, but the researchers suggest that now they do know: because tetracycline targets not one, but two important processes in bacteria. This underscores that multi-target antibiotics have the most potential in clinical use, and it is important to know exactly what mechanisms are present in the different antibiotics known. On top of that, it turns out that membrane-active antibiotics are more effective against persistent bacterial infections. Wenzel and colleagues therefore propose that tetracyclines with enhanced membrane effects should be developed, hopefully contributing to the fight against multi-resistant bacterial strains.
Publication
The findings were published open access in the scientific journal Communications Biology. For the full research article, you can click this link.