Researchers from UvA and Sanquin unravel how white blood cells can crawl through blood vessel walls
13 September 2021
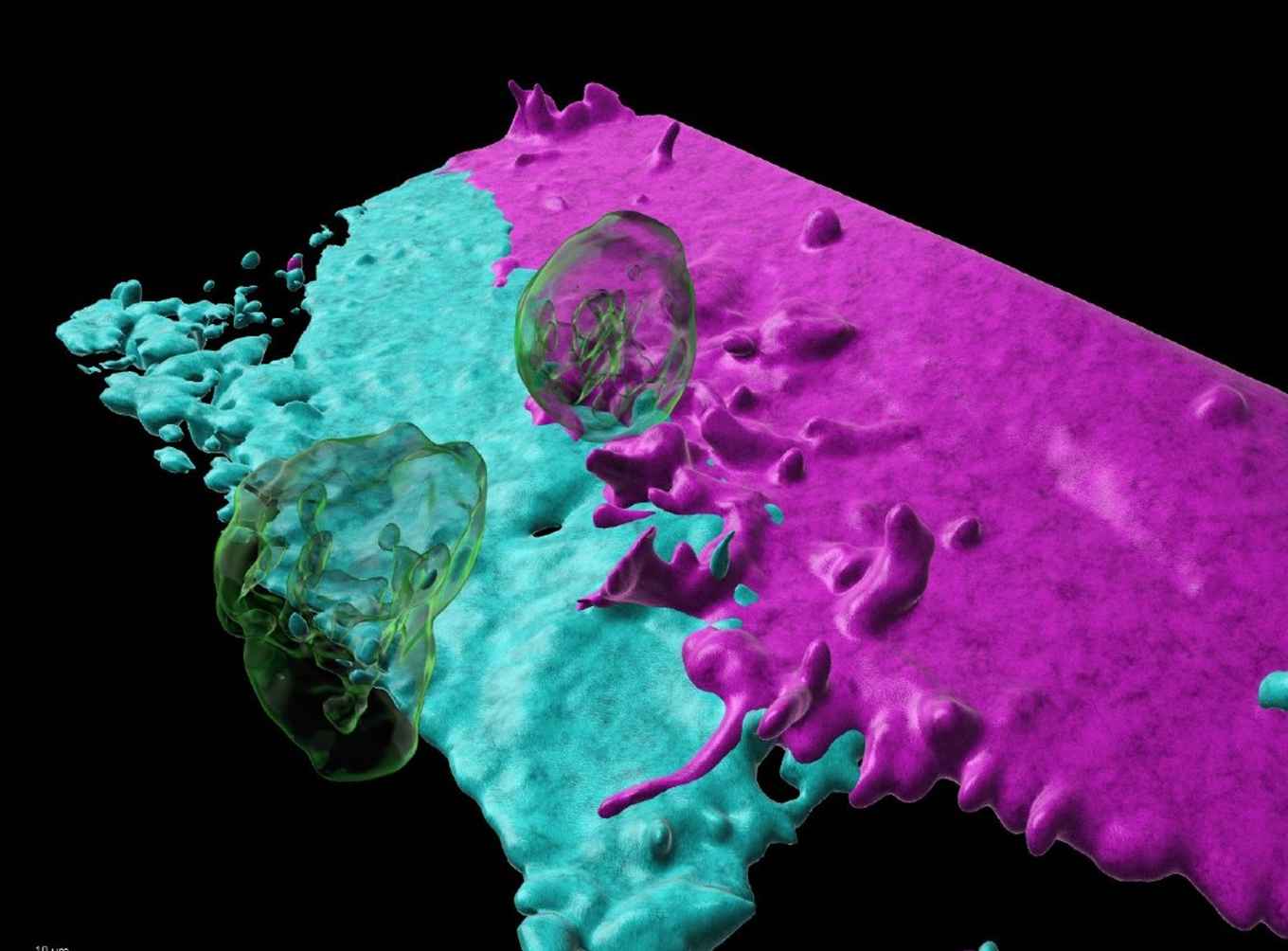
The study builds on long-running research by Van Buul and others into the structure of the walls of blood vessels, and how the blood vessels themselves play a role in whether or not white blood cells can pass through. Van Buul explains: 'We used to think that on the inside blood vessels form a nice, smooth tube. Kind of like a PVC pipe. But it turns out that isn’t always the case. The wall of a blood vessel can also take on a structure similar to sandpaper, to which white blood cells get stuck. You can then see under a microscope that the blood cells start to crawl along this sandpaper, as if they are looking for something. This mainly occurs under inflammatory conditions. They crawl on until they come across a place where a structure similar to a patch of small hairs is present. Those are the preferred hotspots where the white blood cells like to pass through the wall.'
Advanced techniques
This new study, part of the PhD thesis of Janine Arts (UvA & Sanquin) and published in the scientific journal eLife, describes what happens at the location of those hairs. Two state-of-the-art techniques were essential for the research: a very advanced microscope and a molecular “switch”, designed in the laboratory, that allowed the researchers to control the formation of the hairs using light.
'We discovered that the formation of the hairs is controlled by a protein called Rac1,' says Van Buul. ‘UvA team members Eike Mahlandt and Joachim Goedhart have recreated that protein in the lab in combination with a molecular light-sensitive sensor. The sensor ensures that the protein only becomes active if you shine light of a certain wavelength and intensity on it. This allowed us to locally control the production of the hairs on the vessel wall.”
Being able to control the forming of the hairs enabled the biologists to study how white blood cells pass through the vessel wall at the hairy hotspots. This passing through happens fast, and you also want to follow it in 3D, which is hard with a normal microscope. The team therefore went to the US where they could use a so-called lattice light sheet microscope. This advanced microscope is capable of producing high resolution images at a high frequency, that can be combined into a 3D image. ‘It is the first time that anyone has been able to follow this entire process, including the production of the hairs, in such detail,’ says Van Buul.
Potential medical application
The researchers hope that their discovery may in the future help certain groups of patients. Van Buul: 'There are, for example, patients who suffer from chronic inflammation because their body is unable to get enough white blood cells to the site of the infection. We know that in these patients the white blood cells do not stick well enough to the walls of the blood vessels. Potentially, now that we better understand how hairs in the blood vessels form and how white blood cells pass through the vessel wall in these places, it is possible to design drugs that makes this process more efficient in these patients.'
Improving immunotherapy for cancer patients is another potential future application. Van Buul: 'With one of the type of immunotherapy, a patient is administered a large amount of white blood cells in the form of T cells. But the blood vessels in the tumour are often poorly differentiated and do not easily allow those T cells to enter the tumour tissue. The current solution is to administer an extremely high number of T cells to the patient, so that hopefully enough will reach the tumour. But if we could ensure that the blood vessels in the tumour do allow the T cells to pass through, that could also make the therapy more efficient.'
Details of the publication:
Janine JG Arts, Eike K Mahlandt et al, Endothelial junctional membrane protrusions serve as hotspots for neutrophil transmigration, in: eLife 2021, DOI: 10.7554/eLife.66074