Why can we skate on ice?
9 February 2021

As most people have experienced – either by accident or when wearing ice skates – ice is exceptionally slippery. The question why this is the case remains unanswered however, even after 150 years of research on the topic. An important step towards the answer has now been made by physicists from the University of Amsterdam: the slipperiness of ice is caused by highly mobile water molecules at the ice surface. Rinse Liefferink, one of the physicists involved in the project: “The mobility of these surface water molecules enables sliding with low friction. The friction is not always the same: we show in our study that the mobility of the molecules – and therefore the slipperiness of the ice – can be suppressed by increasing the contact pressure or lowering the temperature.”
Not a water layer
It is commonly believed that ice is slippery due to the presence of a thin layer of liquid water on top of the ice surface, which lubricates the sliding contact. The origin of this liquid water, however, remains highly debated. The very first explanation was put forward by John Joly in 1886. He suggested that the local pressure of a skate on the ice results in it melting into liquid water. However, it turned out that this could not be the whole story, as the pressure melting effect would require unrealistically high contact pressures – in particular at ice temperatures below -3.5°C, which are easily reached in frozen canals and on speed skate rinks.
Nowadays, frictional melting is thought to be at the origin of the lubricating water layer: heat generated during the sliding melts some of the ice which lubricates the sliding of the skate on the ice. However, the story does not end there. Liefferink: “As we all know, water can indeed turn a road or a kitchen floor into a highly slippery surface - if the slider is soft, like a rubber shoe or tire, and moves at a high velocity. Ice, in contrast, is also slippery when contacted with the hard surface of an ice skate, and even when that skate is sliding slowly. Therefore, it seems like still something more is happening on the ice surface.”
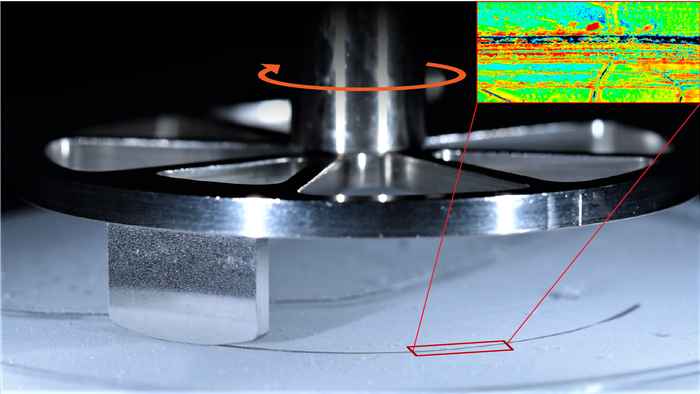
Slippery or not?
In the new study, various types of ice skates were slid over a miniature ice rink while the friction forces were measured. When the temperature was decreased to extreme values as low as -100°C, the resistance to sliding steeply increased, making ice skating impossible. This matched nicely with the results of a previous study, involving some of the same authors: surface water molecules spread over the ice surface in a rolling motion that facilitates the sliding. At -100°C this no longer works. The reason: the mobility of this topmost layer is caused by vibrations of the molecules which become less as they cool down. Only when the temperature is raised these vibrations, and thus the mobility, are large enough to make ice slippery.
It is not just temperature that plays a role, though. In addition, the slipperiness of ice can be removed by increasing the local contact pressure that the skate performs on the ice surface. Due to the chosen shape of the skate and the presence of surface irregularities on the ice and on the skate, the interface where the two touch has many contact points that all contribute to the friction. The sharper the surface irregularities are, the higher the local contact pressure becomes. And when the local pressure at the topmost layer of ice is increased, the resistance to sliding is also significantly increased. This explains an empirically well-known fact: a polished ice skate performs better.
In fact, temperature and pressure work closely together when causing the sliding effect. Close to the melting temperature of the ice, the raised pressure results in ploughing; the skate penetrates the ice and leaves ploughing tracks on the ice surface. This implies that a third factor gets involved: the speed of the skater. Liefferink: “Since both the lubricating ability of the ice surface and the amount of ploughing depend on the sliding speed, the ultimate resistance to sliding is set by an intriguing interplay of temperature, pressure and speed.”
How to skate a new world record?
At the most common speeds and pressures, sliding on ice has the lowest resistance at -7°C – in agreement with the empirically known optimal temperature that ice masters try to achieve at speed skating rinks. At this temperature, the high mobility of the outermost layer of the ice results in a slippery surface where the hardness of the ice can just sufficiently withstand the skate without causing the ploughing effect. At this point, the friction can be further minimized by minimizing the contact pressure of the skate. This is already done by ice skaters: the optimal ice skate has a smooth bottom, hence mostly low pressure and therefore low friction, and sharp edges causing high pressure where necessary for grip. Perhaps the better understanding of why these conventional wisdoms work, can help in setting new world records in the future.
Publication
Rinse W. Liefferink, Feng-Chun Hsia, Bart Weber, Daniel Bonn: ‘Friction on Ice: How Temperature, Pressure, and Speed Control the Slipperiness of Ice’, Phys. Rev. X 11 (2021) 011025.