Prestigious ERC Advanced Grants awarded to five UvA professors
14 August 2013
Awarded projects
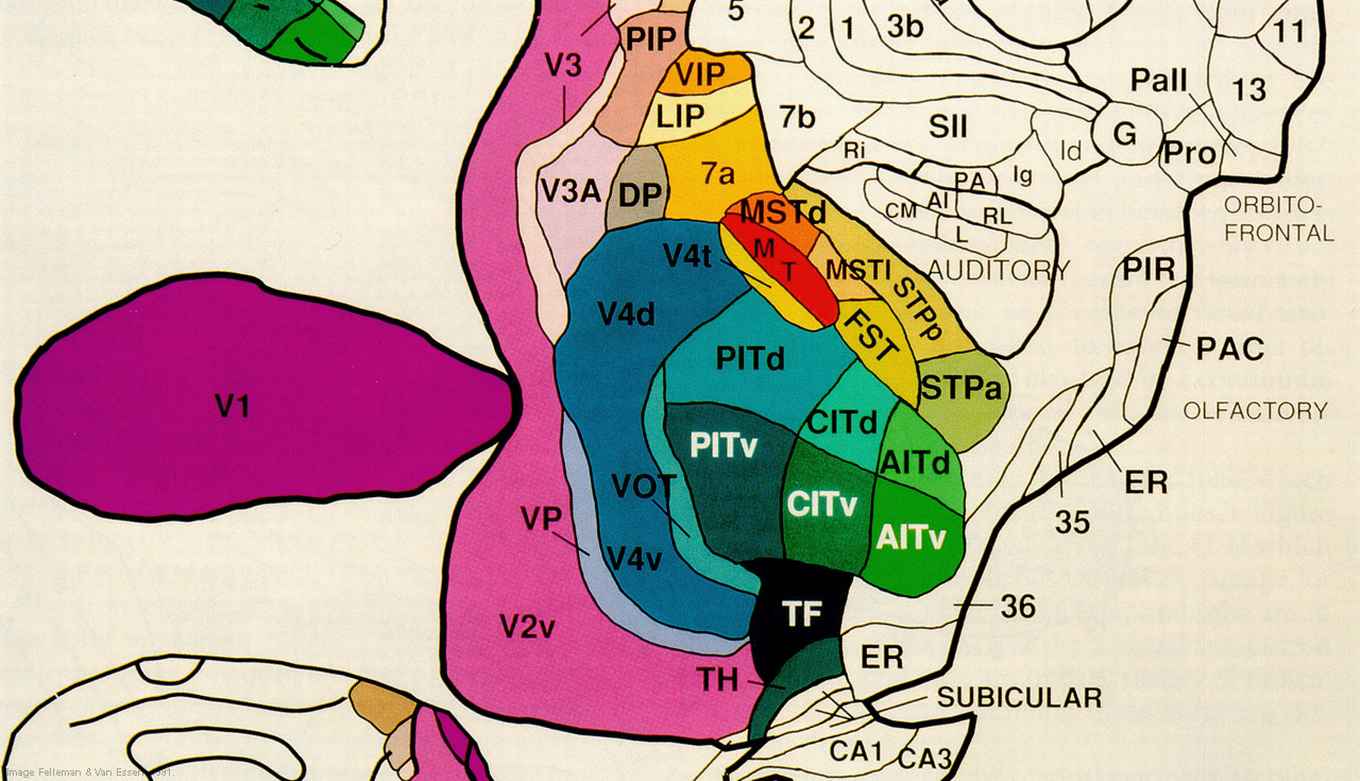
Prof. Edward de Haan, professor of Neuropsychology, Faculty of Social and Behavioural Sciences (FMG): 'A Functional Architecture of the Brain for Vision'
In his research, De Haan aims to develop a new functional architecture of the visual cortex based on two concepts: a methodology for determining the necessity of neural structures for certain partial functions, and a theoretical framework founded on interactive cortical networks. To determine which neural structures are necessary for particular functions (such as identifying colour or motion), he will be studying patients with focal (i.e. localised) brain lesions. This involves research on a large-scale cohort, in conjunction with four academic medical centres throughout the Netherlands. In his research on the manner in which necessary neural structures interact, De Haan adopts a critical view of the 1992 model proposed by Goodale & Milner, the current ‘gold standard’. In their model, Goodale & Milner identify two principal pathways: one for processing the visuospatial information that is used for motor programming, and one for visual recognition and memory. De Haan recently formulated an alternative model, based on the assumption that the neural structures involved are organised into multiple overlapping networks.
Edward de Haan is also the dean of the Faculty of Social and Behavioural Sciences at the University of Amsterdam.
Prof. Joost Reek, professor of Supramolecular Catalysis, Faculty of Science (FNWI): 'Nature Inspired Transition Metal Catalysis'
Metal-based catalysts are crucial to the sustainable production of chemicals in the pharmaceutical and fine chemical industries. Most transition metal catalysts are based on coordination complexes in which the active metal ion is surrounded by ligands (specially designed molecules). Since the activity and selectivity of the catalyst is determined by the interaction between the metal and the ligand, the development of new catalysts has always been dependent on the development of new ligands. This has led to the application of catalysts in a broad spectrum of chemical reactions. With his ERC Advanced Grant, Joost Reek intends to develop new methods of governing the activity and selectivity of transition metal catalysts. To do so he proposes turning to nature, which offers a far greater toolbox for creating selective catalytic systems. In enzymes, the active site is encapsulated by a protein ‘cage’. Using this same mechanism, Reek aims to encapsulate metal complexes (among other things) within synthetic cages, in order to analyse their effects in greater detail. He also plans to develop systems relevant to solar-to-fuel applications, and to the activation of nitrogen. Although such reactions take place efficiently in nature, they are difficult to reproduce in synthetic systems.
Prof. Lex Schrijver, professor of Discrete Mathematics and Optimization, Faculty of Science (FNWI): 'Applying Fundamental Mathematics in Discrete Mathematics, Optimization, and Algorithmics'
Schrijver’s research project aims to strengthen the ties between fields of mathematics that are more fundamental in character (such as algebra, geometry, analysis and topology) and the more recent, applied disciplines of discrete mathematics, optimisation and algorithmics. The overall aim of the project is to design new, effective instruments that can help unravel the complexities of structures such as networks, codes, knots etc. and create a better understanding of such complex structures by means of new characterisations, clearer boundaries and faster algorithms. There have been various developments in recent years involving methods taken from representation theory, invariant theory, algebraic geometry, measure theory, functional analysis and topology that have led to new applications in discrete mathematics and optimisation, both in a theoretical and algorithmic sense. The research results can be applied to areas including networks, coding, routing and timetabling, as well as to statistical and quantum physics, computer science and logistics.
Lex Schrijver is also active as a fellow at the National Research Institute for Mathematics and Computer Science (CWI).
Hergen Spits, Professor of Cell Biology, Academic Medical Center (AMC): 'The Roles of Innate Lymphoid Cells and Rhinovirus in Asthma Exacerbations'
Acute asthma attacks are a major concern, particularly among young children. Their most significant cause is infection with human rhinoviruses (HRVs). Until now, it was thought that type-2 T-helper cells (Th2 - part of the immune system) played a crucial part in this process. By itself, however, Th2 activity only offers a partial explanation. Another type of immune cell (type-2 Innate Lymphoid Cells, or ILC2) seems to play an equally important part. After activation, these cells produce large quantities of specific cytokines, or substances associated with the body’s immune response. Research on mice has shown that this can ultimately lead to asthma. It therefore seems likely that it is not Th2 cells, but ILC2 cells that are the primary cause of acute asthma attacks. Spits suspects that rhinoviruses have either a direct or indirect effect on ILC2. According to his hypothesis, these cells respond by producing cytokines, which can in turn trigger an asthma attack. Spits is focusing on unravelling the mechanisms and function of ILC2 cells following infection with HRV, testing the substances that can affect human ILC2 cells in the lungs, and developing drugs that can attack HRV.
Prof. Rogier Versteeg, professor of Genetics, specialising in Molecular Paediatric Oncology, Academic Medical Center (AMC): 'Challenging the Gaps in Global Cancer Concepts by a Real Life Tumor: Human Childhood Neuroblastoma'
Cancerous cells in a tumour do not form a homogenous mass of identical cells, but instead form a diverse array of heterogeneous cells with unique characteristics that exhibit an ingenious mutual interdependence. Cancer treatments currently aim to kill the cells in the tumour. However, because the tumour contains so many different cell types, this strategy is never completely successful. Although most cancer cells die as a result of treatments such as chemotherapy, other cells in the tumour with different characteristics will survive and cause the cancer to return at a later stage. Versteeg aims to map out the diversity of the cancerous cells within a tumour. He is conducting research on neuroblastoma, a rare form of cancer that affects the nervous system in young children. Although very young children can survive, prognoses are poor for those diagnosed after they reach age 2. Early last year, Versteeg’s research group succeeded in producing a complete genetic map of this type of tumour. With the ERC grant, Versteeg will now focus on the emergence of heterogeneity among cancerous cells in neuroblastoma, as well as on the differences between them and on the pathways via which those differences emerge. The aim is to develop a treatment that can effectively target every cell variation within the tumour, so that patients are cured of the cancer for good.
About the ERC Advanced Grant
The prestigious ERC Advanced Grants are part of the European Seventh Framework Programme (KP7), and are awarded to distinguished academics to support groundbreaking research. In addition to assessing their research ideas, the grant programme also looks at the researchers’ track record and their research environment. For more information, see link below.